FIRESCALE … KNOW MORE!
by Martin Ebbers

- Definitions – Materials and practices
- Scientific Support – New knowledge
- The “Chameleon Test” by Martinus
- Layer Diagram – The polishing phase
- The History – Origins of a fear pattern
Dear Students and Colleagues:
1. Firescale Definitions
“As studio jewelers the term “firescale” on silver – has become ingrained in us. I would like to start with a simple overview of what is involved when we talk about “firescale”: Most jewelers believe, that copper oxides do remain below the surface of silver-copper alloys after annealing and pickling and will haunt them in the polishing stage. This is not correct. Since nobody seemed to differentiate between fine silver surface layers, created while pickling and the base silver alloy, we have this tremendous confusion and fear amongst jewelers. The word “Firescale” is used for at least two very different metal behaviors and appearances, reaching from mild polishing problems to extreme conditions that result in the breakdown of a silver alloy’s cohesiveness. Let’s keep it simple while looking at basic facts and processes used in our studios.”
- Many shape changes; rolling, drawing, bending and forging harden the inner crystal structure of our alloy.
- Annealing is essential to line up the crystals to soften the inner consistency of our metal in order to continue working.
- As a side effect of annealing and soldering, the surface layer of our metal transforms, creating the conditions for discoloration to occur.
Sterling and Oxides
While heating our work, first reddish, then grayish and later black oxides appear on the surface as a result of the 7.5 % copper content (added for strength in Sterling silver). As a consequence, we need to acid treat (or “pickle”) our silver work. During “pickling”, copper oxides are dissolved and leached out from the silver alloy’s surface, leaving the piece with a matte white, spongy, pure silver top layer. Multiple annealing or soldering will increase the layers of pure silver. It is important to understand that these layers will finally loose all of their copper content and cannot oxidize anymore! In fact, these spongy silver layers are the cause of our polishing difficulties. It is important to know that the Sterling metal below remains unaffected by oxides under regular studio conditions. The base can be polished beautifully if the fine silver is removed! This is not the same as in the extreme case: where the Deep Layer Oxidation is a consequence of severe overheating. In Deep Layer Oxidation the oxides proceed into the alloys interior and polishing becomes futile. “Firescale” seems to have become synonymous with troubles and blamed for all of the above.
back to top
2. Scientific Support
In February of 2012 three polished test plates with typical discoloration were shipped to the Technology Department at the ARGEN Corporation, a highly sophisticated, precious metals Company in San Diego. With a series of x-ray tests they determined the precise surface structure of our sterling silver test plates and helped with understanding the metals’ behavior.
The previously held belief of oxides being the cause of silver discoloration is clearly IRRELEVANT for studio jewelers. Our expanded knowledge is as complex as it is surprising! Simply explained, the surface of copper-depleted sterling silver demonstrates a “Chameleon Effect”. This happens as the metal is trying to re-establish a balance between copper and fine silver at its surface layer. According to Argens’ Scientists, heat in the form of friction from the polishing wheel, acts as a catalyst compelling the copper atoms to rise and re-bond with fine silver atoms on the surface. This re-bonding is evident in the micro-thin discolored surface areas on the polished silver. A second application of higher heat by flame will completely reverse this process, as the copper descends downward and a pure white silver surface remains. (see images: 5,7, and 9).
back to top
3. The “Chameleon Effect”
In order to help his students find clarity and rely on their own observations, Martinus developed the following test. It can easily be duplicated by studio jewelers. As the combined behavior of fluxes and acids with heat has not been thoroughly examined to date, the following series of images explores the firescale discoloration and describes it precisely.

(1) Clean Shapes: A sterling silver plate with an evenly polished surface and no discoloration. (Left: A leaf shape to be soldered onto the plate).
 (2) Fluxing (start): A borax-based flux is applied to the leaf shape.
(2) Fluxing (start): A borax-based flux is applied to the leaf shape.
 (3) Flux (early stage): The Flux protection is spotty, red cuprous oxides appear.
(3) Flux (early stage): The Flux protection is spotty, red cuprous oxides appear.
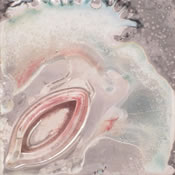 (4) Soldering: Higher temperatures cause the flux to spread out absorbing trapped oxidation. The unprotected areas are developing dark cupric oxides.
(4) Soldering: Higher temperatures cause the flux to spread out absorbing trapped oxidation. The unprotected areas are developing dark cupric oxides.
 (5) Acid Bath or Pickling: Acid has dissolved flux and the copper oxides, which are now leaving an irregular pattern of pure silver on the surface. – The Chameleon Effect, stage one.
(5) Acid Bath or Pickling: Acid has dissolved flux and the copper oxides, which are now leaving an irregular pattern of pure silver on the surface. – The Chameleon Effect, stage one.
 (6) Polish: Heat energy is generated by the polishing wheel, compelling copper atoms to rise from the sterling base.
(6) Polish: Heat energy is generated by the polishing wheel, compelling copper atoms to rise from the sterling base.
 (7) Discoloration: Chameleon Effect, stage two: After polishing, we see that the same former white, pure silver areas, (# 5), have become reddish-grey copper surface areas.
(7) Discoloration: Chameleon Effect, stage two: After polishing, we see that the same former white, pure silver areas, (# 5), have become reddish-grey copper surface areas.
 (8) Reheating: Chameleon Effect, stage three: With heat applied the effect reverses, the copper layer recedes into the sterling silver leaving a white, frosty, pure silver “bloom”.
(8) Reheating: Chameleon Effect, stage three: With heat applied the effect reverses, the copper layer recedes into the sterling silver leaving a white, frosty, pure silver “bloom”.
 (9) Observation: The reddish areas of image 7 turned white and the sterling oxidation helps us in defining the trouble zones. Note, the solutions are not far from here!
(9) Observation: The reddish areas of image 7 turned white and the sterling oxidation helps us in defining the trouble zones. Note, the solutions are not far from here!
Conclusion:
Clearly deep oxides are not the cause of polishing difficulties; instead we have copper atoms transitioning between the surface and lower layers. In short, if we could polish our pieces without generating heat, the word ‘firescale’ would not exist for jewelers. Martinus tested this theory by “cold polishing” a silver test plate connected to a cooling steel block with 1000 grid emery paper. Afterward polishing compounds, (Tripoli & Rouge), were applied gently by hand – the conclusion is: discoloration does not appear during a cold polish.
A second test was conducted by applying a buffing wheel to the previously “cold polished” surface. And there it was, the discoloration came right away: The Argen conclusion is correct; when heat is applied, a copper-leached, pure silver surface wants to revert back to its original structure and causes copper atoms to rise from the Sterling base below. We discovered that it is the fine silver layer next to the sterling base that affects the discoloration problem. Painfully, this happens at the worst possible time, as we are trying to complete our pieces!
back to top
4. Layer Diagram
Unfortunately, it is not easy to illustrate cold polishing. Alternatively, the following two images can help us understand how and where the copper rises. We are using a polishing wheel and Tripoli to penetrate three, spongy, fine silver layers. These had each been established after annealing and pickling. The lowest fine silver layer always attracts copper atoms to rise via friction from the sterling underneath. This layer is very thin but extremely resistant, before we finally can reach and break through to the sterling base, where polishing is easy.

(10) Sterling Silver Test Plate & Diagram of Cross Section
A = fine silver “bloom”, B to D = three layers of copper depleted fine silver, E = copper rising through friction, F = Sterling base
What can we do to make use of our new knowledge?
Once Sterling’s behavior is understood, you will have to choose one of two options.
See Bench Tips on our website!
back to top
5. History of a fear pattern
Throughout work related literature, I have found only vague descriptions and insufficient translations, mostly stressing the rare and worst case scenarios; deep oxidation and the destruction of the physical integrity of a piece, to be seen in the following images where really, polishing becomes useless.

(#11) Firescale, the extreme under the microscope: On the lower side, we see crusty oxidation after hours of over heating in a laboratory. Oxygen enters the alloy deeply. Internal oxidation occurs while copper & silver crystals become isolated, loosing connectivity.
Deep Layer Oxidation (see #11 microscopic cross section in a laboratory test and #12 a hollow ware piece, both Allgemeine Refinery Germany). Here in hollow ware work extreme heat and multiple picklings result in the loss of malleability and ultimately the breakdown of the metal’s cohesiveness. Through temperature related expansion and silver’s high receptivity and storage capacity to oxygen when heated, the copper and silver crystals run into a deepening loss of connectivity through copper oxidation towards the interior of an alloy. At times, we may find these unwanted deep effects in reticulation as well.

(#12) Firescale, frequent annealing and pickling have caused deep oxidation, cracks open in the forging of this hollow ware piece
Usually, we deal with mild forms of difficulties which I am really reluctant to refer to as Firescale! For clarity we may be better off to speak of Thin Layer Oxidation, see image #13 (Allgemeine Refinery Germany) and from earlier on, Martinus #1 to #7. This second type of oxidation doesn’t affect the integrity of our metal. At present, we do not have qualified, scientific information which also takes into consideration all of our daily studio procedures and so I decided to contact the highest authority on the matter, Master Goldsmith, Scientist and Author, Professor PhD. Erhard Brepohl in Germany.

(#13) Correctly annealed sheet silver up to a light glowing, shows no interior oxidation, the top and bottom layer of a 925 silver alloy displays the typical Thin Layer Oxidation. Here’s where all of our common polishing difficulties originate!

(#7) Sudden discolorations in the polishing phase, related to Thin Layer Oxidation. Copper is rising to the surface.
In March 2011 he invited me to discuss “Firescale” and discoloration of Sterling silver at his house in Bad Doberan. I encountered a brilliant scholar and a generous man with a good sense of humor. Brepohl seemed perplexed that surface discoloration during the polishing phase would cause distress for North American jewelers. He emphasized that his research regarding deep layer oxidation is the result of extreme laboratory conditions and does not apply to ordinary studio procedures. In the 1950s, Professor Brepohl lead extensive metallurgic research involving specialized laboratory conditions while masterminding the theoretical foundation for all the later jewelry-making books. We agreed that “firescale”, according to his own research and as defined in the McGraw-Hill Dictionary, is a rare case scenario and usually does not relate to our work as studio jewelers. Also admittedly these definitions are not stating such differences, which unfortunately led to misinterpretations and unquestioned translations have created unnecessary fears amongst jewelers.
While in agreement, that Fine Silver layers are the true vehicle of the issue, Professor Brepohl offers two unconventional suggestions to avoid difficulties in the polishing phase:
First: We can choose to minimize the fine silver layer by acid treating our work at the very end, only once. Oxidation would become our surface protection during assembly.
Second possibility: Once assembly is complete, we have the option of dipping our piece briefly, for a few seconds in 10 % Nitric Acid to dissolve the fine silver layer prior to polishing. Be aware of hand and eye protection, fumes are also hazardous!

Prof. Brepohl & Martinus
Bad Doberan, Germany
March 2011
Introductory quote from the highly esteemed resource book – “The Theory and Practice of Goldsmithing”:
“The mutual exchange of experience is an important means of further education. Those who allow their colleagues to participate willingly in their own experiences will often get useful information in return.”
Professor PhD. Erhard Brepohl
Please see Martinus` Bench Tips for further advice!

